A metallized coating (a metallic alloy applied to a base metal or concrete) is intended, in most applications, to be a form of protection to an underlying metal substrate. The act of applying the coating is referred to as metallizing. Metallizing can be achieved in several ways such as hot-dipped galvanizing or thermal arc spraying, applied in situ or in a shop. Zinc, which was first used in construction in 79 AD, is the most used metal for this process. Half of the zinc produced today is used for corrosion protection of steel.
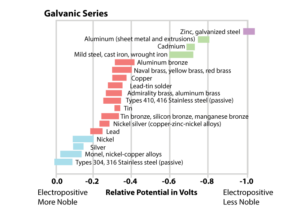
Zinc is the prominent metal of choice for metallized coatings as it corrodes slower than ferrous metals and is less noble than steel. Noble metals are resistant to chemical actions and oxidation and do not corrode. In the Galvanic Series of Metals, metals are ranked from ‘noble’ to ‘active’ based on the metals’ potential. Zinc is often a good choice as an anode due to its electronegative potential (Figure 1).
In electropotential relationships, when two metals are joined, the more electronegative metal becomes anodic to the more noble metal. At this state, the anodic metal corrodes preferentially to the more noble metal. This is commonly referred to as sacrificial corrosion, where the anodic metal sacrifices itself to the cathodic metal. (See Table 1 which demonstrates the corrosion performance of zinc and zinc-based alloys in contact with other metals, as an indicator of bi-metallic suitability. In most instances, zinc corrodes preferentially to the metals listed.)
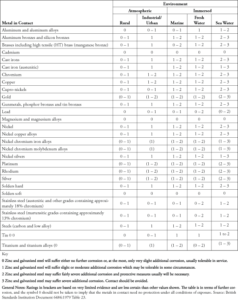
Table 1. As presented in Commentary of Corrosion on Bi-metallic Contacts. Originally referenced as “Table 23 Additional Corrosion of Zinc and Zinc Based Alloys resulting from contact with other Metals or Carbon”.
The range of metallized zinc coatings can be seen in Figure 2. This image depicts the types and thickness of the applied metal to the substrate. These coatings are generally termed as galvanized coatings, though their application methods differ.
The focus of this article is to look at metalized coatings applied to steel and concrete where the coating is used to prevent corrosion and extend the service life of the treated component.
Metallized Coatings
Metallized coatings are applied to the underlying substrate thermally or by arc-flame spraying. The metal is in the form of a wire which is melted and sprayed onto the surface by compressed air. Once the liquefied metal hits the surface, it cools rapidly and forms a cohesive coating.
Applications directly onto metal surfaces are performed with either zinc or an aluminum alloy. The metallized coating provides corrosion resistance by acting as a barrier and a sacrificial metal. Figure 2 illustrates a comparison of the various zinc coatings in cross section.
Applications are carried out in the field or in the shop, dependent on the type of component. Where components are too large for hot dip galvanizing, metallizing is the first-choice alternative. Applications in the field are numerous and generally fall under the following categories:
- Extending the Life of Existing Galvanized Components (Zinc)
- Alternative to Painted Component, where corrosion resistance is required (Zinc or 85:15 Alloy)
- Concrete Surfaces for Corrosion Control and Cathodic Protection (Zinc or Al/Zn/In) and Reinforcing Steel
The primary benefit of metallizing is long-term increased durability, particularly in salt-rich environments. Here, metalizing provides a whole life cost advantage over more traditional methods of protection which tend not to perform as well in harsh environments. Entire bridges, which are subjected to salt-rich environments, are prime candidates for metallizing, as are bridge components which are subjected to drainage and runoff.
Galvanized Components
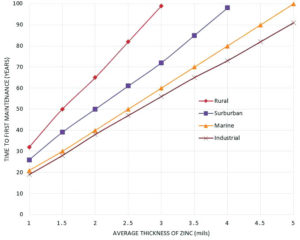
The long term corrosion resistance of zinc coatings is mainly dependent on coating thickness and the environment to which the coating is exposed. When both of these are known, then the time to first maintenance can be determined as seen in Figure 3. This often has a direct correlation on maintenance and life cycle costs. Time to first maintenance is defined as the time it takes for 5% of the steel surface to rust. Coatings are measured in mils, as seen in Figure 3. One (1) mil is equivalent to 25.4 microns (µm) or 0.56 ounces per square foot.
One of the main benefits of galvanizing is the metallurgical bond between the zinc and the underlying steel or iron which creates a barrier that is part of the metal itself. The bond between the zinc and steel is approximately 3,600 pounds per square inch (PSI) which is six (6) times better than bond strengths for conventional coatings which fall between 300 and 600 PSI.
In addition to the bond, galvanizing provides abrasion and impact resistance due to the formation of the zinc-iron layers. Four layers are formed: Gamma, Delta, Zeta and Eta, where each has a different hardness. The Gamma, Delta, and Zeta layers have a hardness higher than the underlying metal. This hardness provides abrasion resistance and excellent protection against coating damage. The Eta (top) layer is quite ductile, providing an element of impact resistance as well as an element of self-healing.
Performance data on galvanized coatings dates back to 1926. The American Society of Testing Materials (ASTM) Committee A05 (Metallic-Coated Iron and Steel Products) has been collecting records of performance based on various atmospheric conditions. Atmospheric conditions are extremely variable, reducing coating life considerably in some instances. Factors affecting coatings can be defined as follows:
- Wind Direction
- Corrosive Fumes
- Pollutants
- Amount of Salt Spray
- Number of Wetting and Drying Cycles
- Duration of Moisture Exposure
Applications of galvanizing are extensive. Structural members of bridges and buildings, electrical utility poles, and artistic structures are commonly treated. Component sizes vary considerably from a small nut or nail to a bridge girder support.
In summary, the major benefits of galvanizing are:
- Factory Quality Assurance (QA) Controlled
- Complete Coverage
- Superior Bond to Steel
- Hardness of Coating Compared with Steel
- Excellent for Indoor or Outdoor Environments
When galvanized coatings lose Zn, they can be metallized to extend their service life.
Alternative to Painted Components
The most commonly used alternative to painting steel is the application of a metalized coating in the form of zinc/aluminum or 85/15 alloy, 85% zinc, and 15% aluminum by weight. Both options provide a metal coating with superior corrosion resistance over standard paint coatings. Metallizing can be used to cover welds, seams, ends, and rivets to improve corrosion resistance and can be applied up to 10 mils (254µm) thick.
In a testing program supported by the Federal Highway Administration (FHWA), zinc and 85/15 alloy applied at 6 mils outperformed over 40 other coatings. The coatings were exposed for seven (7) years in a harsh marine environment with and without a sealer top coat. These panels showed virtually no corrosion and no deterioration from intentional coating defects at the end of this exposure period.
In 2012, the Bureau of Reclamation carried out a laboratory evaluation of metallized coatings for use on reclamation infrastructure. Five thermal spray alloys and two sealers were investigated using laboratory test methods that included immersion, accelerated weathering, adhesion, and electro-impedance spectroscopy.
In summary, the Bureau determined, by simulating environments, the best use of metallizing would be on radial gates, partially exposed trash racks, and other equipment subjected to a fluctuating immersion environment. The Bureau also determined metallizing to be beneficial in severe atmospheric service environments such as bridges and above-ground piping.
In their test program, the pure aluminum was believed to have offered the best combination of protection in immersion or fluctuating immersion, as long as the water had a pH between 4.0 and 8.5.
Concrete Corrosion Control and Reinforcing Steel
Corrosion of reinforcing steel can have detrimental effects on the service life and functionality of a concrete structure. In structures that have high chloride exposure, significant chlorides at the reinforcing steel depth can trigger steel corrosion even in the presence of a highly alkaline concrete. Chloride ion ingress from sea salt, de-icing salt, or contaminated concrete mix constituents breaks down the passive oxide layer without damaging the concrete. Once there is sufficient concentration at the steel surface, along with sufficient oxygen and moisture, corrosion initiates.
Author’s note: The high pH of new concrete passivates the steel. The formation of an oxide layer (Fe2O3) or Magnetite protects the steel from corrosion until such time corrosion initiates. As concrete material conditions degrade, by age, through exposure to the elements, construction defects, or other related causes, the steel is no longer protected and is in an environment where it can corrode.
In the corrosion reaction, iron dissolution occurs at the anode and two electrons (2e–) are gained elsewhere on the steel to maintain electrical neutrality. The gain of electrons at the cathode reaction results in the formation of alkaline hydroxyl ions at the steel surface. This reaction is harmless to the steel and the surrounding concrete. Thus, if this reaction is forced to occur by adding current, then the steel is protected. Damage does not occur if the cathodic reactions are controlled; hence, the term cathodic protection.
For this type of corrosion control, an ‘anode’ is installed or applied to a structure and provides current (electrons). In 1992, the Federal Highways Administration was quoted by the Strategic Highway Research Council (SHRP-C/UPW-92-618) stating that “cathodic protection is the only method known to stop corrosion damage regardless of the level of chloride within the structure.” Two types of cathodic protection are available today: Galvanic and Impressed Current. Galvanic Cathodic Protection (GCP) is based on the Galvanic Series of Metals where the electronegative metal gives up electrons to the electropositive metal. Impressed current cathodic protection (ICCP) utilizes an external DC power source connected to the anode system to provide current.
Metallized coatings, thermally sprayed (TS) onto concrete, can provide suitable anodes for cathodic protection and corrosion control in concrete. Figures 4 through 9 illustrate application and testing of a TS coating of a bridge pier. TS anodes can be used in either galvanic or impressed current mode, depending upon the circumstances, conditions, and environment near the structure. The types of anodes suitable for a TS application for cathodic protection include:
- Zinc (Zn) (GCP/ ICCP)
- Aluminum/ Zinc/ Indium (A-Z-I) (GCP/ ICCP)
- Titanium (ICCP only)
Early comparative thermal spray anode performance trials performed by the California Department of Transportation concluded that thermal/flame-sprayed zinc provided the best combination of cost, effectiveness, acceptability, and coating consumption rate for cathodic protection. TS Zn coating types have a long history of reducing life cycle costs of reinforced concrete bridges on a national and international level. Aside from mitigating corrosion through cathodic protection, the zinc retards migration of the chlorides to the reinforcing bar, and re-alkalization occurs at the steel and concrete interface during the cathodic reaction.
The efficacy and service life of TS alloys on concrete is based on several parameters:
- Sufficient Surface Preparation
- Adequate & Uniform Coating Thickness
- Current Density
- Cumulative Charged Passed
- Acidification
- Pore Solution of Concrete
- Environmental Interactions
- Moisture
- Precipitation
- Wetting and Drying
- Bond Strength
The electronegative potential of Zn allows for it to function well as an anode in galvanic mode in a moist environment. Al/Zn/In alloys include the use of Indium to activate the Aluminum and have been designed for use where moisture is less abundant.
The Oregon DOT has been successfully employing TS to Oregon’s historic coastal highway system. Their bridge inventory includes over 120 bridges, most of which are reinforced concrete. Over 430,566 square feet of bridge surface area have been treated using thermal-sprayed (TS) zinc anodes in both impressed current and sacrificial cathodic protection (CP) systems.
The Florida DOT’s experiences mirror that of ODOT, whereby studies have shown that anode consumption in a galvanic mode is largely dependent upon moisture and bond. FDOT also found that if metallized coatings are sprayed directly onto spalls where the reinforcing is exposed, then corrosion activity is slowed, providing a longer duration time between repairs.
For galvanic use, the service life of the alloy is based on coating thickness and current density resulting in anode consumption. This is largely a function of moisture and chloride exposure. Bridge substructures in marine environments are highly suitable for GCP systems; however, splash zones on bridge piers may require a more robust approach and drier components may be more appropriate for an ICCP system. Both the Oregon DOT and Florida DOT studies identified that if sufficient bond strength is achieved between the concrete substrate and anode, anode service life in ICCP systems may exceed 25 years.
Like traditional cathodic protection systems, TS applied systems have permanently embedded reference electrodes to monitor system performance. This allows for depolarization tests to be performed so that the National Association of Corrosion Engineers (NACE) and British Standards European Norm (BSEN) criteria for cathodic protection can be demonstrated. The successful use of TS systems has also been documented in parking garages, pre-stressed structures, post-tensioned structures, and bridges in non-marine environments exposed to high chlorides.
In 2000, Bennett, et. al, published their work on the use of metallized zinc alloys on a post-tensioned garage in Pennsylvania exposed to deicing salts, a substructure of the M4 Motorway in the United Kingdom exposed to chloride runoff (deicing salts), and a condominium complex facing the Atlantic Ocean situated on Marathon Key, Florida. These studies illustrate the efficacy and success of the TS Zn anodes in a variety of environments, applications, and structure types. All case studies presented achieved the 100mV depolarization criteria required by the NACE Standard SP 208 and BSEN 12696:2012, illustrating the suitability and broad use of TS coatings for corrosion control.
The purpose of the TS applied alloys is to provide a service life extension to both the structure and the concrete repairs, by decreasing the deleterious effects of corrosion to the surrounding concrete and by reducing repair cycles and maintenance costs.
Recently published NACE International Standard SP0216-2016, Sacrificial Cathodic Protection of Reinforcing Steel in Atmospherically Exposed Concrete Structures is now available.
Conclusions
Metallized coatings comprise a relatively limited range of alloys applied to a steel or concrete surface to provide greater durability for the service life of the structure or component. These applications can include: 1) batch galvanizing of Zn to steel such as nuts, bolts, and even reinforcing steel, 2) metallizing exposed steel structures, and 3) surface applying alloys to reinforced concrete bridges, garages, and buildings to serve as anodes in cathodic protection systems. Metallized coatings provide a significant service life extension to the treated part or system. Their flexibility in a TS application allows for a variety of uses on complex geometries as well.
The service life of the coating depends on a multitude of factors, all of which are largely environmentally driven. Therefore, consideration for the service environment must be addressed prior to material selection. Additionally, the success of the metallized coating is dependent upon proper surface preparation of the metallic or concrete substrate, and adequate bond strength between the two materials. Whether galvanizing or metallizing, the service life extension of the steel components can be in the range of 25 years before the first maintenance is required. This makes the use of metallized zinc and associated alloys a cost effective, low maintenance material choice for barrier protection, self-healing, cathodic protection, and managing chloride ingress into a substrate.▪
Definitions
Whole Life Costing (WLC) is “an economic assessment considering all agreed projected significant and relevant cost flows over a period of analysis expressed in monetary value. The projected costs are those needed to achieve defined levels of performance, including reliability, safety, and availability”. Source: BS/ ISO 15686-5 Buildings & Constructed Assets.
Life Cycle Costs (LCC) are the cost of an asset, or its part throughout its cycle life while fulfilling the performance requirements. (BS/ ISO 15686-5 Buildings & Constructed Assets: Service Life Planning: Life Cycle Costing.)
References
Corrosion Barrier Coatings, one-year Evaluation of Marine Coating Exposures, U.S. Navy, David W. Taylor Research and Development Center, 1985.
Laboratory Evaluation of Metalized Coatings for Use on Reclamation Infrastructure – Technical Memorandum No. MERL-2012-14
Grantham. Concrete Repair: A Practical Guide. Spon Press: Routledge: Taylor Francis Group. London. 2011.
Broomfield and Tinnea. Strategic Highway Research Council. Cathodic Protection of Reinforced Concrete Bridge Components. FHWA-SHRP-C/UPW-92-618. National Research Council. 1992.
Bennett, Bushman, Costa, Noyce. Field Applications of Performance Enhancing Chemicals to Metallized Zinc Anodes. NACE Corrosion 2000. Paper No. 00790.
ODOT Research Unit, Albany Research Center, USDOE, and Tinnea Associates, and FHWA. Intermittent Application of Cathodic Protection; Interim Report. SPR# 317. Washington DC 2005.
NACE International. SP0290-2007 (formerly RP0290), Impressed Current Cathodic Protection of Reinforcing Steel in Atmospherically Exposed Concrete Structures.